Introduction
Asphyxiation is a state of insufficient oxygen supply to the body. While it typically is caused by low oxygen concentration in air it can occur in normal oxygen concentrations, if there is an impairment to the oxygen transportation system, such as in carbon monoxide poisoning. Asphyxiant gases can be divided into chemical asphyxiants (such as carbon monoxide and hydrogen sulfide) which have a direct toxic effect, and simple asphyxiants (such as nitrogen, helium, and argon) which are not directly toxic to the human body. Simple asphyxiants, such as inert gases, cause asphyxiation by reducing oxygen concentration in air and in severe cases can cause death [1,2].
In Korea, suicides using asphyxiants have rapidly increased since 2008, following a celebrity death by carbon monoxide poisoning [3,4]. Argon, an inert gas used in welding, can be obtained relatively easily and inexpensively [5,6]. Although in Korea there have been reports of domestic suicide attempts using inert gases, attempts using argon gas are rare. Therefore, the authors report the first experience of suicide attempts using argon gas in Korea with a review of the literature.
Case
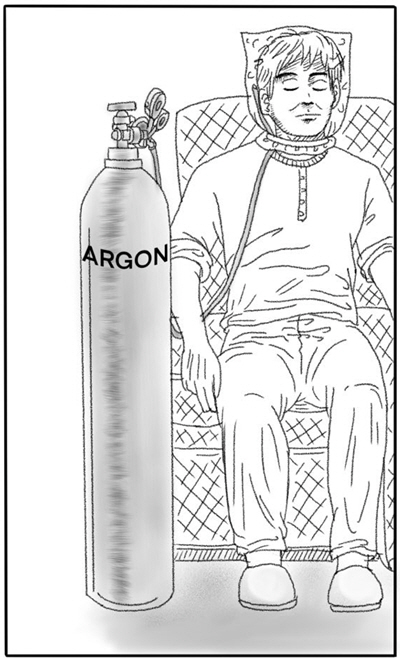
A 24-year-old man, with a history of depression, was brought into the emergency room with impaired consciousness. On arrival, his vital signs were as follows: blood pressure, 140/90 mmHg; pulse rate, 145 beats/min; respiratory rate, 29 breaths/min; body temperature, 37.4°C; oxygen saturation (using a non-rebreather mask), 97%. He was drowsy, Glasgow Coma Scale (GCS) 13/15, the pupils were equal in size (3 mm) and reactive to light. According to his parents, they had left the house for about one hour and returned to find their son next to a cylinder of argon gas (Fig. 1). He was seated in a chair with a plastic bag over his face which was connected to the cylinder by a rubber pipe. There was condensation on the inside of the bag, and the mouth of the bag had a string that could be tightened around the neck to prevent gas leakage (Fig. 2). A suicide note and a receipt for the purchase of argon gas were found near the patient.
Initial, arterial blood gas analysis showed: pH, 7.265; pCO2, 34.0 mmHg; pO2, 101.4 mmHg; bicarbonate, 12.1 mmol/L; base excess, –10.6 mmol/L; lactic acid, 9.1 mmol/L; methemoglobin, 0.2% and carboxyhemoglobin, 0.5%. Blood tests showed: white cell count, 8,571/uL; hemoglobin, 13.6 g/dL; sodium, 134 mmol/L; potassium, 4.1 mmol/L; chloride 91 mmol/L; aspartate transaminase (AST), 42 U/L; alanine transaminase (ALT), 21 U/L; blood urea nitrogen (BUN), 11.0 mg/dL; creatinine, 0.85 mg/dL and glucose, 102 mg/dL. The patient was administered oxygen at 12 L/min via a non-rebreather mask, and normal saline infusion was commenced. Computed tomography of the brain was performed to exclude other causes of impaired consciousness; no acute lesions were observed. After three hours, the patient’s GCS score was normal (15/15), and he had a residual mild headache. Arterial blood gases and lactate levels normalized. The patient revealed he had attempted suicide following a deterioration in his depressive symptoms. He had learned about this method of suicide, and purchased the argon gas online. He was discharged without complications 2 days.
Discussion
Although inert gases are considered safe and easy to handle as they are unreactive, they can act as simple asphyxiants. These gases are typically defined as Group 18 (VIIIa) in the periodic table, and consist of helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Nitrogen may also be considered an unreactive gas [5,6]. The incidence of suicide by inert gas asphyxiation is increasing worldwide, and there have been several reports in Korea [7-9].
Argon is the most abundant inert gas on Earth, accounting for around 0.93% of the Earth’s atmosphere. It has a molecular weight of 39.95 g/mol, which is heavier than air (28.8 g/mol), and its specific gravity relative to air is 1.35 [5]. Argon is stable at high temperatures and is commonly used in steel and iron manufacturing, welding, and cutting. It is colorless and odorless making it difficult to detect. While it is not by itself toxic, in enclosed spaces asphyxiation can occur, as its higher density relative to air causes oxygen displacement [5,6]. According to Yoo et al. [10], eight workers suffered asphyxiation injuries due to argon gas while working in enclosed spaces, between 1999 and 2007 in Korea.
Studies show that suicide attempts using argon are less common than those using helium or nitrogen. Azrael et al. [11] analyzed suicides in the United States between 2005 and 2012, and reported that 4% of deaths were caused by gas inhalation, of which carbon monoxide was most common (73%), followed by helium (21%), hydrogen sulfide (1%), and nitrogen (1%). Gunnell et al. [12] reported that of 2,495 suicide cases using gas between 2001 and 2011 in England, there were three deaths due to argon. Yau and Paschall [13] investigated 968 suicides, using chemical substances or gas between 2005 and 2014, and found six caused by argon gas poisoning; fewer than those caused by helium or nitrogen. However, experimental studies have reported that argon is a stronger asphyxiant than helium or nitrogen. Altland et al. [14] discovered that rats that had been exposed to helium, nitrogen, or argon showed survival rates after one hour of 92%, 60%, and 12% respectively, demonstrating that argon was the strongest asphyxiant. Another study identified that argon gas has a sedative effect via actions on GABAA, the receptor targeted by benzodiazepines [15]. Thus, argon could present a more attractive option for those considering suicide as it may help alleviate fear. In Korea, argon gas is inexpensive and easily accessible online without any regard for intended use, so more concerns are arising.
The normal concentration of oxygen in air is 21%. Following argon gas inhalation, manifestations of oxygen deficiency appear when oxygen concentration drops below 16%; these include quickening of the pulse and respiratory rate, vomiting and headache. At oxygen concentrations below 10%, the patient may experience a loss of consciousness, seizure, and a dramatic decrease in pulse, ultimately resulting in death by asphyxiation. Prolonged resuscitation, beyond six minutes, could result in severe neurological sequelae [6,16]. The treatment priority is to establish a rapid and plentiful oxygen supply, which may include mechanical ventilation [6]. In this case the patient’s suicide attempt was not successful, probably due to incomplete sealing of the plastic bag allowing outside oxygen to enter, and a fairly short duration of exposure to the argon.
Suicide using inert gas first gained public awareness in 2002, when Derek Humphry’s suicide manual, “Final Exit”, described a method involving helium and a plastic bag [17]. This ‘pain-free method’ has since spread indiscriminately on suicide websites. In an analysis of suicides involving inert gases in Korea, all cases used a plastic bag (Table 1). Lim et al. [8] analyzed 17 suicides using helium. The mean age was 30.6 years, with ten cases aged 20–29 years, three aged 30–39 years, and four aged 40–49 years. Younger individuals, who may be more familiar with the Internet, have easier access to suicide information online. Korea takes pride in its status as a world leader in information technology, though easily obtained suicide information could increase the risk of inert gas suicides. Prevention strategies must include strict monitoring of suicide websites and the introduction of robust systems for checking the identity and qualifications of those purchasing inert gases.